I. What Is Fabric Water Vapor Permeability?
Fabric water vapor permeability refers to a fabric’s ability to allow water vapor to pass through it, and it is one of the core indicators for evaluating the comfort of functional textiles. During physical activity, the skin continuously evaporates moisture (sweat). If the fabric cannot promptly expel this water vapor, a hot and humid microenvironment will form on the skin’s surface, leading to discomfort.
Water vapor permeability and water repellency together constitute the dual standards of “breathability” and “protection” for outdoor functional apparel. An excellent piece of functional apparel must both block the intrusion of external liquid water and rapidly expel water vapor from the body—this is precisely where the value of water vapor permeability research lies.
II. Testing Methods for Water Vapor Permeability
1. Desiccant Method (Positive Cup Method)
Standards: GB/T 12704.1, ASTM E96, JIS L1099 A-1
Principle: A desiccant (anhydrous calcium chloride) is placed in a moisture permeability cup, and the test sample is sealed at the cup opening. After being kept in a constant temperature and humidity environment for a specified period, the moisture permeability is calculated by measuring the weight gain of the desiccant.
Features: The test conditions are mild and closely resemble actual wearing conditions, making it the most commonly used testing method in China.
2. Evaporation Method (Water Method, Inverted Cup Method)
Standards: GB/T 12704.2, ASTM E96 BW, JIS L1099 B-1
Principle: A specific volume of distilled water is placed in a moisture transmission cup. After sealing the sample, the cup is inverted. Water vapor passes through the sample and evaporates outward; the moisture transmission rate is calculated by measuring the decrease in water mass within the cup.
Features: Testing conditions are relatively harsh, simulating a high-humidity environment, making it suitable for waterproof and breathable fabrics.
3. Sweating Guarded Hotplate Method
Standards: ISO 11092, GB/T 11048
Principle: A porous metal plate is used to simulate skin perspiration. The energy required to maintain a constant temperature is measured, directly yielding the fabric’s moisture resistance (Ret) and water vapor transmission index (imt).
Features: This method more closely approximates actual human physiological conditions and is considered the “gold standard” for evaluating thermal and moisture comfort.
III. Key Factors Affecting the Moisture Permeability of Fabrics
(Ⅰ) Fiber Raw Material Factors
1. Moisture Absorption of Fibers
The moisture absorption of fibers is the foundation for determining moisture permeability. Based on how fibers interact with water, they can be classified as follows:
- Hydrophilic fibers: cotton, viscose, wool, silk, etc. The molecular structure of these fibers contains hydrophilic groups (-OH, -COOH, -NH₂), which can adsorb water molecules via hydrogen bonding and transport them through diffusion within the fiber. Fibers with strong moisture absorption typically exhibit better moisture vapor permeability.
- Hydrophobic fibers: Synthetic fibers such as polyester, nylon, and polypropylene. These fibers do not absorb water themselves; water vapor is primarily transported through the pores in the gaps between fibers. Although the moisture vapor transmission capacity of a single fiber is limited, excellent moisture vapor transmission performance can be achieved through specialized structural designs.
2. Fiber Fineness and Cross-Section Shape
- Fiber fineness: The finer the fiber, the larger the specific surface area, and the more dense the capillary network formed between fibers, which facilitates the conduction of water vapor. Fabrics made from ultra-fine fibers (<0.5 dtex) typically exhibit superior moisture vapor transmission performance.
- Cross-sectional Shape: Fibers with non-circular cross-sections (such as cross-shaped, trefoil-shaped, or hollow fibers) can form more pore channels compared to circular fibers, thereby increasing the pathways for water vapor transmission. For example, hollow polyester fibers not only reduce weight but also significantly enhance moisture permeability and thermal insulation.
(II) Yarn Structure Factors
1. Yarn Twist
Twist directly affects the density and porosity of the yarn:
- Low-twist yarn: The yarn is loose, with large gaps between fibers, resulting in low resistance to water vapor transmission and good moisture permeability. However, the yarn has lower strength and reduced abrasion resistance.
- High-twist yarn: The yarn is compact, with reduced gaps between fibers, leading to increased resistance to water vapor transmission and reduced moisture permeability. However, the yarn has high strength, and the fabric feels stiff and crisp.
2. Yarn Blend Ratios
- Cotton/Polyester Blends: Cotton fibers provide moisture absorption and wicking capabilities, while polyester provides strength and quick-drying properties. When the cotton content is between 40% and 60%, moisture permeability and durability achieve a good balance.
- Wool/Nylon Blends: The natural moisture-absorbing properties of wool combined with the strength of nylon are commonly used in high-end outdoor apparel.
(III) Fabric Structure Factors
1. Fabric Density and Tightness
- Increased Density: Warp and weft yarns are arranged more tightly, reducing fabric porosity, increasing resistance to water vapor transmission, and lowering moisture permeability.
- Decreased density: Fabric porosity increases, improving moisture vapor permeability, but this may lead to reduced wind resistance and insufficient strength.
2. Fabric Thickness
Fabric thickness is inversely correlated with moisture vapor permeability: the diffusion resistance that water vapor must overcome to pass through the fabric is directly proportional to its thickness.
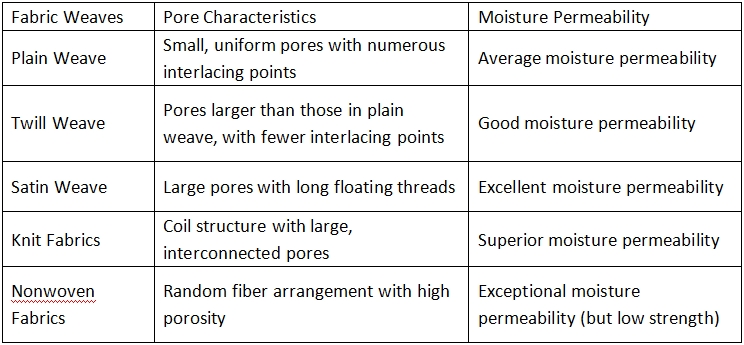
3. Weave Structure
The pore morphology formed by different weave structures varies significantly:
4. Multi-layer Composite Structures
Modern functional apparel often employs multi-layer composite designs:
- Two-layer fabrics: The outer layer is windproof and waterproof, while the inner layer wicks away moisture and sweat; the “pump effect” created by the pores between the layers enhances moisture vapor permeability.
- Three-layer composites: Fabric + waterproof and breathable membrane + lining. The membrane’s microporous structure allows water vapor molecules to pass through while blocking liquid water.
(IV) Post-processing Factors
1. Coating Treatments
Coating is a key method for modifying moisture vapor permeability:
- PU Coating: Standard PU coatings block fabric pores, significantly reducing moisture vapor permeability. However, microporous PU or hydrophilic PU coatings can maintain a certain level of moisture vapor permeability while retaining waterproofing properties.
- Microporous PTFE Membrane: Features extremely high porosity and very small pore sizes, offering high moisture vapor transmission rates and water pressure resistance.
- Non-porous hydrophilic membranes: These utilize a “adsorption-diffusion-desorption” moisture transmission mechanism via hydrophilic groups on the molecular chains. While their moisture permeability is slightly lower than that of microporous membranes, they offer better wash resistance.
2. Water-repellent and moisture-permeable finishing agents
- Fluorocarbon compounds: These impart water- and oil-repellent properties to fabrics but have a certain negative impact on moisture permeability.
- Silicone-based compounds: Form a breathable film on the fiber surface, with minimal impact on moisture permeability.
- Nanotechnology treatments: Such as nano-SiO₂ and TiO₂, which impart special functions while maintaining porosity.
3. Moisture-Wicking Finishes
Treating hydrophobic synthetic fiber fabrics with hydrophilic finishing agents (such as polyether-based or polyacrylic-based compounds):
- The finishing agents form a hydrophilic layer on the fiber surface, improving surface wettability.
- A wicking effect is created, rapidly transporting sweat from the skin side to the outer layer for evaporation.
- Moisture vapor transmission can be increased by 20%–40%, while maintaining the quick-drying properties of synthetic fibers.
(V) Environmental Factors
Both moisture vapor transmission testing and actual use are significantly influenced by environmental conditions:
1. Temperature
- Rising temperature: The kinetic energy of water vapor molecules increases, accelerating diffusion; simultaneously, fibers expand after absorbing moisture, enlarging their pores. Moisture vapor transmission rate increases significantly with rising temperature.
2. Relative Humidity
- High-humidity environment: The concentration gradient of water vapor on both sides of the sample decreases, reducing the driving force and lowering the moisture vapor transmission rate.
- Low-humidity environments: The concentration difference is large, resulting in high moisture vapor transmission rates.
- Isothermal and isohygric conditions: When the temperature and humidity on both sides of the sample are identical, the moisture vapor transmission rate is zero.
3. Wind Speed
- Increased wind speed accelerates the loss of water vapor from the sample’s surface, maintaining a higher concentration gradient and thereby increasing the moisture vapor transmission rate.
- In actual outdoor wear, wind speed is a key variable affecting perceived comfort.
IV. Balancing Moisture Permeability with Other Functions
1. Moisture Permeability vs. Water Resistance
Conflict: Water resistance requires extremely small or closed fabric pores, while moisture permeability requires open pores.
Solutions: Microporous membrane technology, hydrophilic non-porous membranes, and high-density weaving techniques (such as high-density fabrics made from ultra-fine fibers).
2. Moisture Permeability vs. Windproofing
Conflict: Windproofing requires a tightly woven fabric structure, while moisture permeability requires unobstructed pores.
Solutions: Multi-layer composite structures, membrane technology, and adding a windproof fabric layer on the outer side of the moisture-permeable membrane.
3. Moisture Permeability vs. Thermal Insulation
Conflict: Thermal insulation requires a layer of still air (loose structure), but excessive thickness reduces moisture permeability.
Solutions: Hollow fibers, 3D knitted structures, and phase-change materials for temperature regulation.
Conclusion
Fabric moisture vapor permeability is a complex system involving multiple interacting factors, spanning disciplines such as fiber science, textile engineering, polymer materials, and heat and moisture transfer.
UTSTESTER provides precise moisture vapor permeability testing equipment. If you encounter any issues during moisture vapor permeability testing, please feel free to contact our technical team at any time.

Email: hello@utstesters.com
Direct: + 86 152 6060 5085
Tel: +86-596-7686689
Web: www.utstesters.com